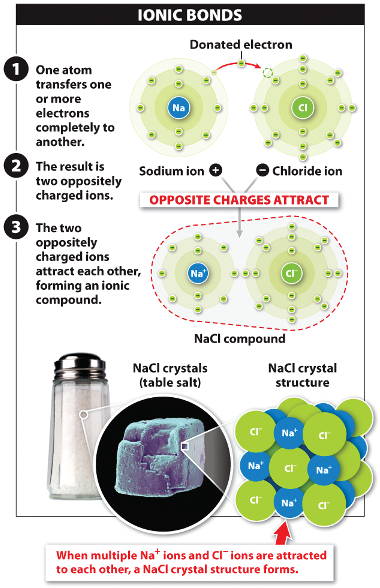
 Ionic bond
Ionic bond
- A sodium
atom (Na)
can donate
an electron to a chlorine
atom
(Cl).
- The positively charged sodium
ion and the negatively charged chloride
ion
are attracted to each other.
- Sodium chloride (NaCl, or table salt)
is composed of alternating
sodium
and chloride
ions
held together by strong ionic bonds.


 Ionic bond
Ionic bond